 
It is a white caustic alkaline crystalline solid at room temperature. Like sodium oxide, calcium nitride absorbs. It reacts with water or even the moisture in air to give ammonia and calcium hydroxide: 4 Ca 3 N 2 + 6 H 2 O 3 Ca (OH) 2 + 2 NH 3. It can be produced by direct reaction of the elements: 3 3 Ca + N 2 Ca 3 N 2. 
Is CaO a lime Calcium oxide (CaO) commonly known as quicklime or burnt lime is a widely used chemical compound. Calcium nitride is formed along with the oxide, CaO, when calcium burns in air. The reaction gives a beautiful flame by performing a Flame test on Ca. Since Calcium oxide is ionic we need to balance charges when we write the formula. This makes calcium a positive ion with a charge of 2+. The reaction for the preparation of calcium oxide is shown below. The calcium oxide is prepared from the thermal decomposition of limestone which contains calcium carbonate. It follows that calcium and oxygen can achieve their octet states by reacting with each other: calcium atoms donate two of its electrons to oxygen atoms, in contrast to Forming molecular compounds. The ionic form of calcium oxide is given as Ca2+O2 C a 2 + O 2. Calcium oxide molecules are made up of one calcium cation (with a charge of +2) and one oxygen anion (which holds a charge of -2). O, oxygen (happi) gains the octet state by receiving two electrons.Ca, calcium (kalsium) gains the octet state by donating two electrons, and. 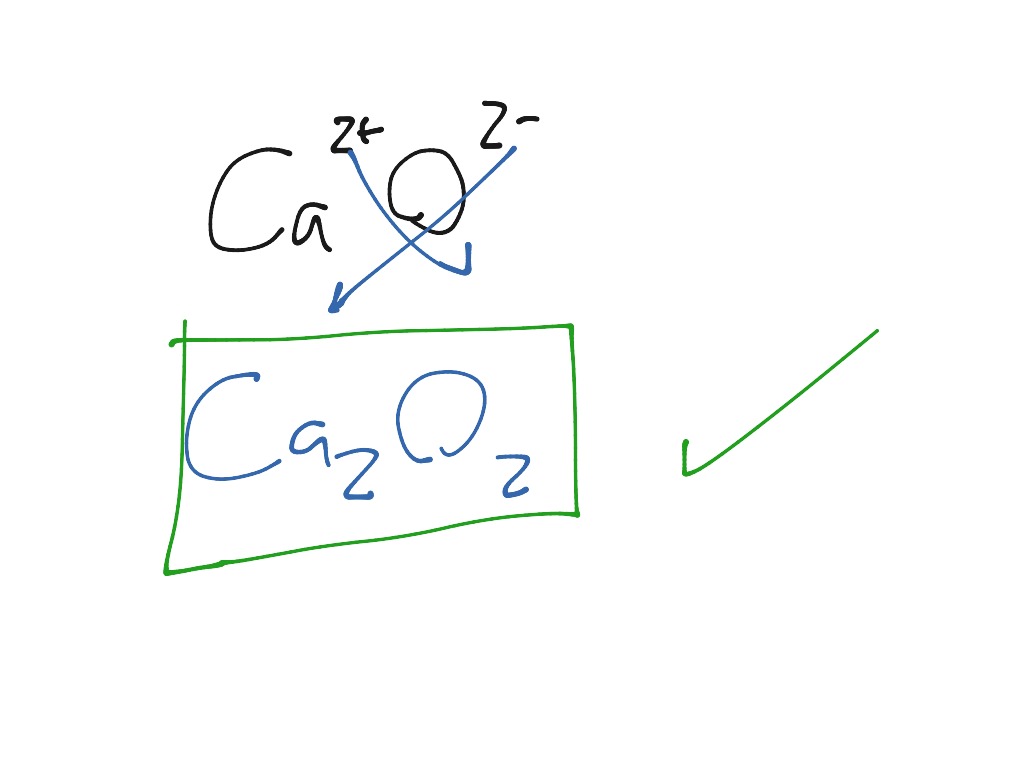
Calcium peroxide or calcium dioxide is the inorganic compound with the formula CaO 2. Using the logic behind the periodic table studied in detail in From bonds to reactions, In summary, one formula unit of calcium hydroxide (Ca(OH)2) contains two hydroxide ions and one calcium ion with a charge of +2. Strontium peroxide Barium peroxide Sodium peroxide: Except where otherwise noted, data are given for materials in their standard state (at 25 ☌ 77 ☏, 100 kPa). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
